You are here
About the Philadelphia Chromosome
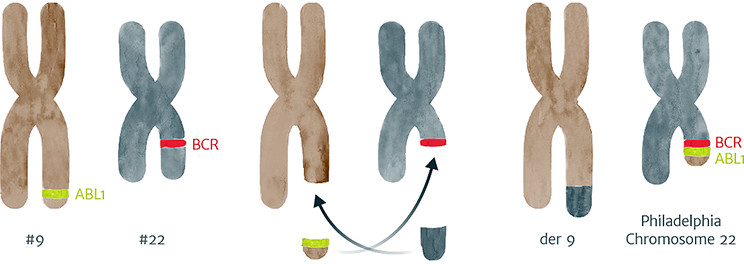
It is formed when chromosomes 9 and 22 swop one part each with the other. It is not yet understood what causes this to happen. CML is not an inherited condition and as such cannot be passed on to children.
Known as a reciprocal translocation, a piece containing the ABL1 gene from the bottom part of chromosome 9 breaks off and attaches to a region on chromosome 22 where the BCR gene is located, thus forming a new chromosome 22 containing the abnormal fusion gene BCR-ABL1
The new shortened chromosome 22 is called the Philadelphia chromosome. You might also see it written as t(9; 22). The BCR-ABL1. fusion gene makes a protein called a tyrosine kinase. These kinds of proteins are located on or near the surface of cells and send signals that speed up cell division.
Ph+CML is a rare disease with an incidence of 1 to 1.5 new cases per 100.000 people each year. It is very rare in young people under 19 and ultra-rare in young children.
In approximately 95% of cases, the Ph chromosome is detected by cytogenetic analysis. In around 5% of the remaining suspected cases, the Ph chromosome is not visible by cytogenetic tests. However, the BCR-ABL1 fusion gene can be identified by molecular testing with q-PCR in approximately half of those remaining 5% of cases. For simplicity, any disease that contains the BCR-ABL1 fusion gene is referred to as Ph+ CML. If you do not have the Ph chromosome or evidence of the BCR-ABL1 gene then you do not have CML.
It has been shown that the BCR-ABL1 fusion gene is the single definitive marker for Ph+ CML and remains the key abnormality throughout the chronic phase of the disease.
The BCR-ABL1 fusion gene translates as a protein tyrosine kinase, referred to as Bcr-Abl1. The activity of tyrosine kinases is typically controlled by other molecules, but the abnormal tyrosine kinase produced by the BCR-ABL1 gene means the signal for cell division is always ‘switched on’. This continuous signalling activity sets up a cascade of events that allows for unregulated cell division (cancer). Over time the Ph+ cells grow to overpopulate the marrow, crowding out normally functioning cells.
Since the introduction of targeted therapy with tyrosine kinase inhibitors (TKIs), it is increasingly evident that, at least in chronic phase, when cells containing the BCR-ABL1 gene are reduced to very low molecular levels, the risk of disease progression is significantly reduced.
Note*: Genes are denoted with italicised capitals as in ABL1; BCR etc., whereas proteins are denoted with non-italicised capitals, sometimes followed by lower case, as in Bcr-Abl1.